Expanding access to monoclonal antibody-based products: a global call to action


<p>A roadmap to making lifesaving monoclonal antibody treatments affordable and available, particularly to those living in low- and middle-income countries. </p>
Monoclonal antibodies (mAbs) are one of the most powerful tools in modern medicine. From cancer to COVID-19, they are being developed to treat many different diseases. However, most of the world’s population don’t have access to them.
This report sets out a roadmap to making these lifesaving treatments affordable and available, particularly to those living in low- and middle-income countries.
- what monoclonal antibodies are and how they are used to treat different diseases
- an analysis of the global monoclonal antibody market, including the barriers to access
- a roadmap of the actions that need to be taken to make equitable access a reality.
- researchers
- global health community
- pharmaceutical and biotech companies
- funders
- regulators
- policy makers
Wellcome's affordable innovations for global health flagship supported IAVI to produce this report.
Monoclonal antibody development is one of the fastest-growing segments of biomedical research
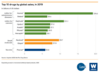
- In 2019, seven of the 10 best-selling novel drugs were mAbs for cancer and inflammatory diseases.
- More than 570 mAbs are currently in clinical testing.
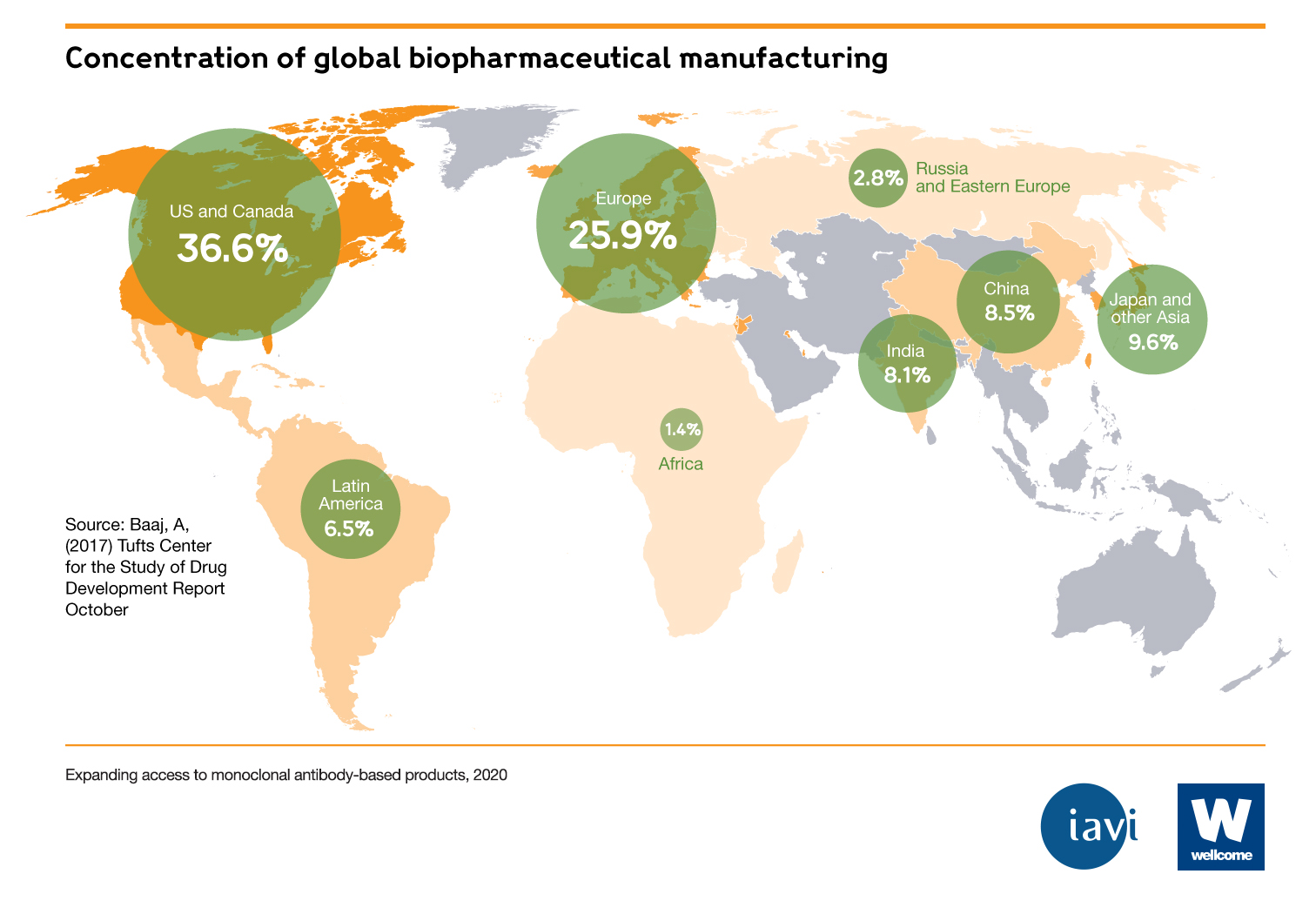
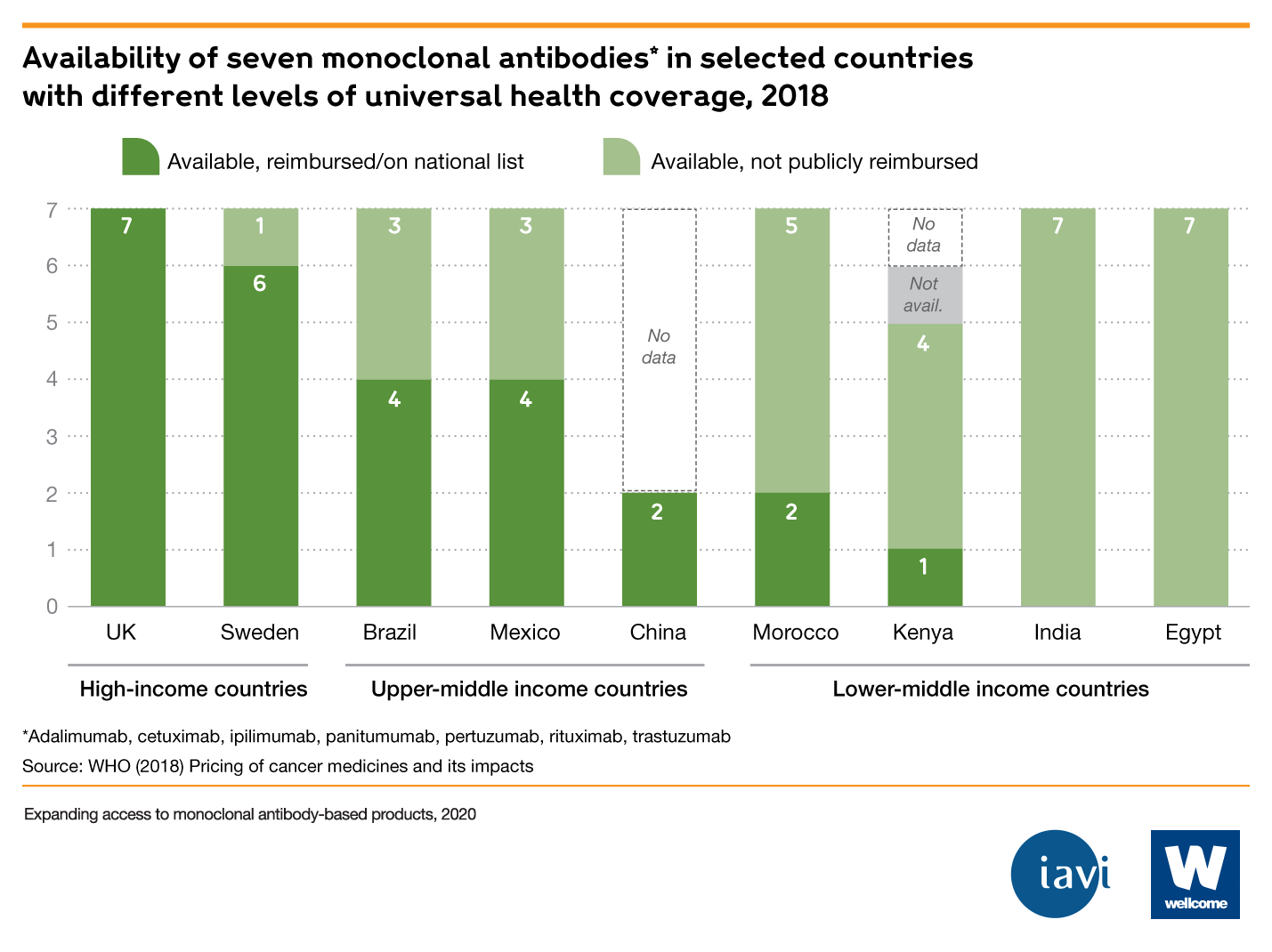
There is a global need for monoclonal antibodies, but they are not globally accessible
- 80% of mAbs are sold in the United States, Europe and Canada. Only 20% of mAbs are sold in the rest of the world, which makes up 85% of the global population.
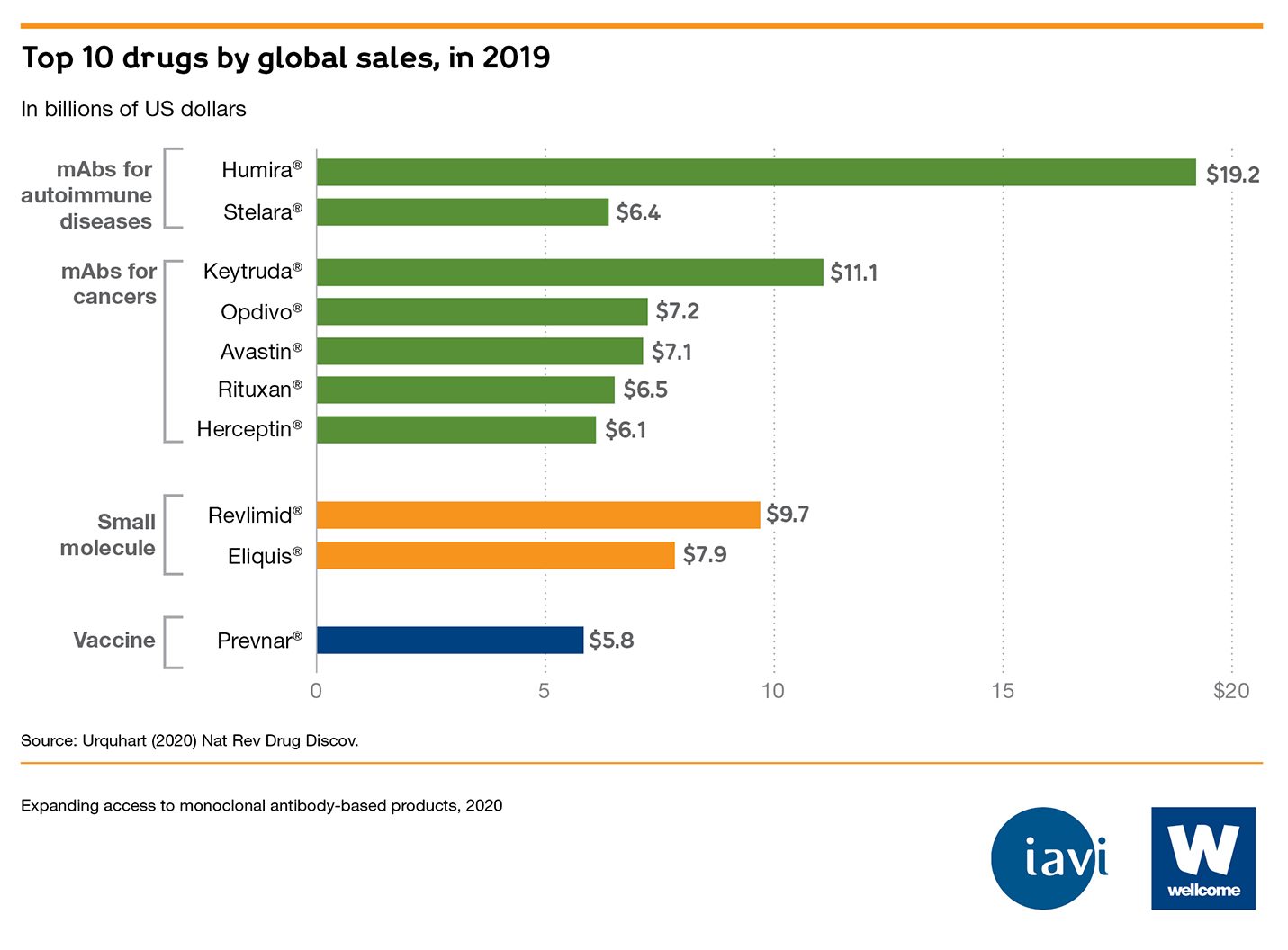
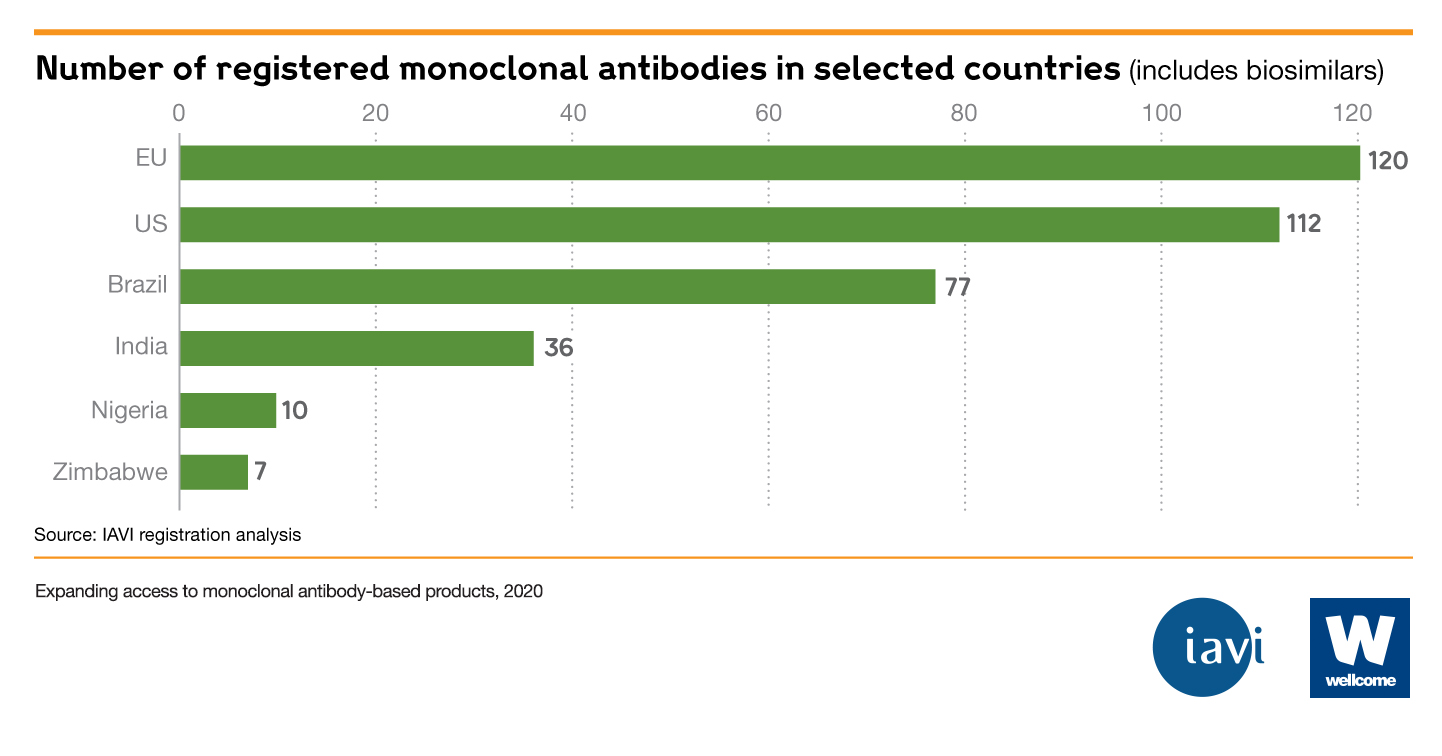
- Few, if any, mAbs are available in low- and middle-income countries (LMICs). India – which has the most registered mAbs for a LMIC – has 36, compared to the US (112) and Europe (120).
- Monoclonal antibodies are among the world's most expensive treatments. For example, the median price for mAb treatment in the United States is $15k-200k per year. Where mAbs are available in LMICs, they often aren’t reimbursed by public health systems, which adds to their unaffordability.
We need to take action to reshape the monoclonal antibody world
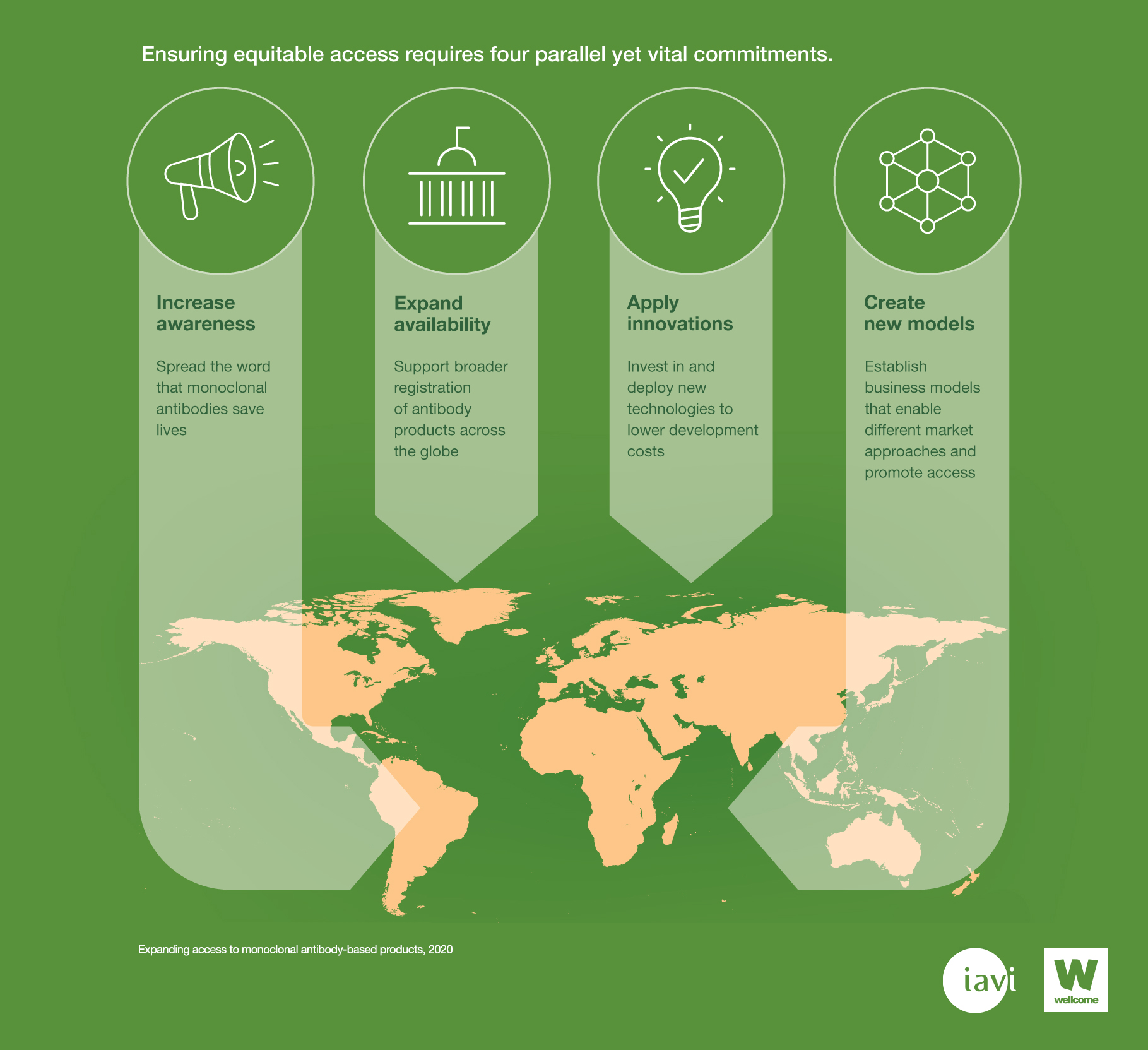
Ensuring equitable access to monoclonal antibodies requires four parallel commitments:
- Increase awareness: spread the word that monoclonal antibodies save lives
- Expand availability: support broader registration of monoclonal antibodies across the globe
- Apply innovations: invest in and deploy new technologies to lower development costs
- Create new models: establish business models that enable different market approaches and promote access.












- For more information, contact Lindsay Keir at l.keir@wellcome.org.


Innovations Flagships support the development of exciting new products, technologies and other interventions to prevent or treat disease.


We want to help the world overcome Covid-19. Research is the best way to save lives – but more investment in accessible treatments, tests and vaccines is urgently needed to change the course of the pandemic.

