Expanding access to monoclonal antibody-based products: a global call to action
Listing image

File size
267975 bytes
Image width
1808px
Image height
1106px
Filename
monoclonal-antibody-market-is-unbalanced_0.png
Image

Download allowed
On
Listing summary
[Summary] A roadmap to making lifesaving monoclonal antibody treatments affordable and available, particularly to those living in low- and middle-income countries.
Priority areas
Teams
English
Topics
Standfirst
Monoclonal antibodies (mAbs) are one of the most powerful tools in modern medicine. From cancer to COVID-19, they are being developed to treat many different diseases. However, most of the world’s population don’t have access to them.
This report sets out a roadmap to making these lifesaving treatments affordable and available, particularly to those living in low- and middle-income countries.
Content
Admin title
Download the report
Group
Link
Link style
Primary
Content
Body
- what monoclonal antibodies are and how they are used to treat different diseases
- an analysis of the global monoclonal antibody market, including the barriers to access
- a roadmap of the actions that need to be taken to make equitable access a reality.
Body
- researchers
- global health community
- pharmaceutical and biotech companies
- funders
- regulators
- policy makers
Body
Wellcome's affordable innovations for global health flagship supported IAVI to produce this report.
Group title
Key findings
Admin title
Key findings
Group
Body
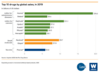
Monoclonal antibody development is one of the fastest-growing segments of biomedical research
- In 2019, seven of the 10 best-selling novel drugs were mAbs for cancer and inflammatory diseases.
- More than 570 mAbs are currently in clinical testing.
There is a global need for monoclonal antibodies, but they are not globally accessible
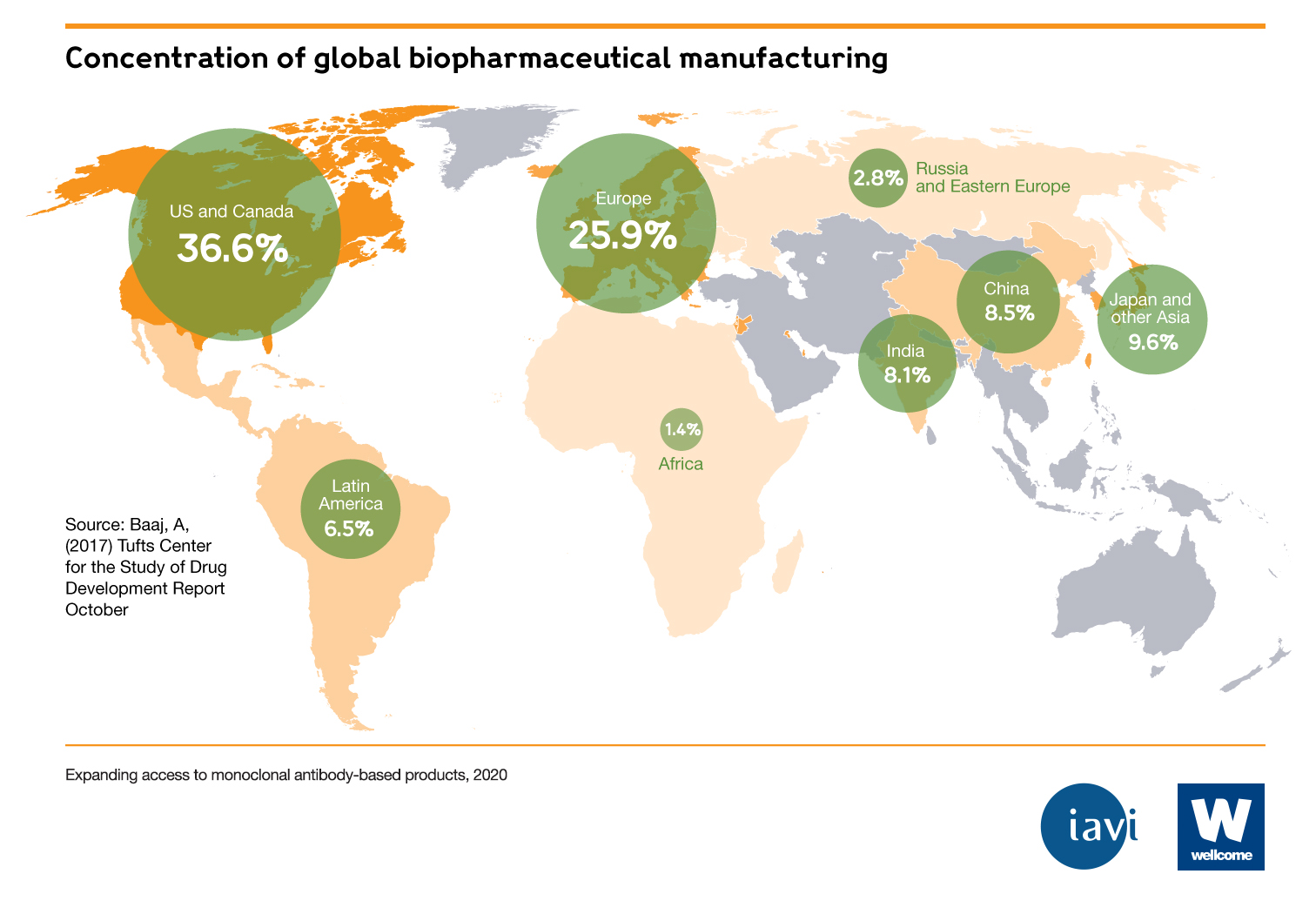
- 80% of mAbs are sold in the United States, Europe and Canada. Only 20% of mAbs are sold in the rest of the world, which makes up 85% of the global population.
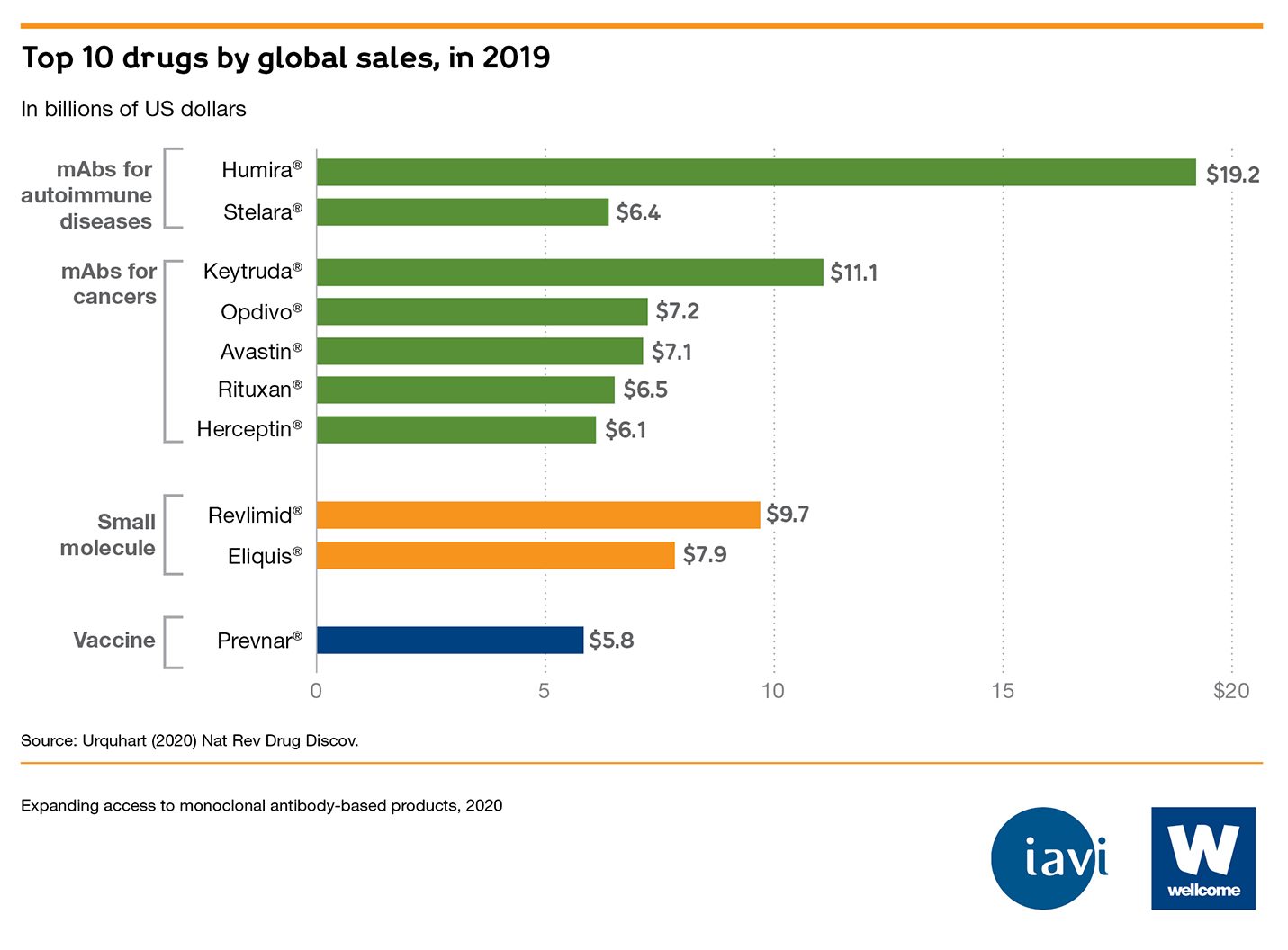
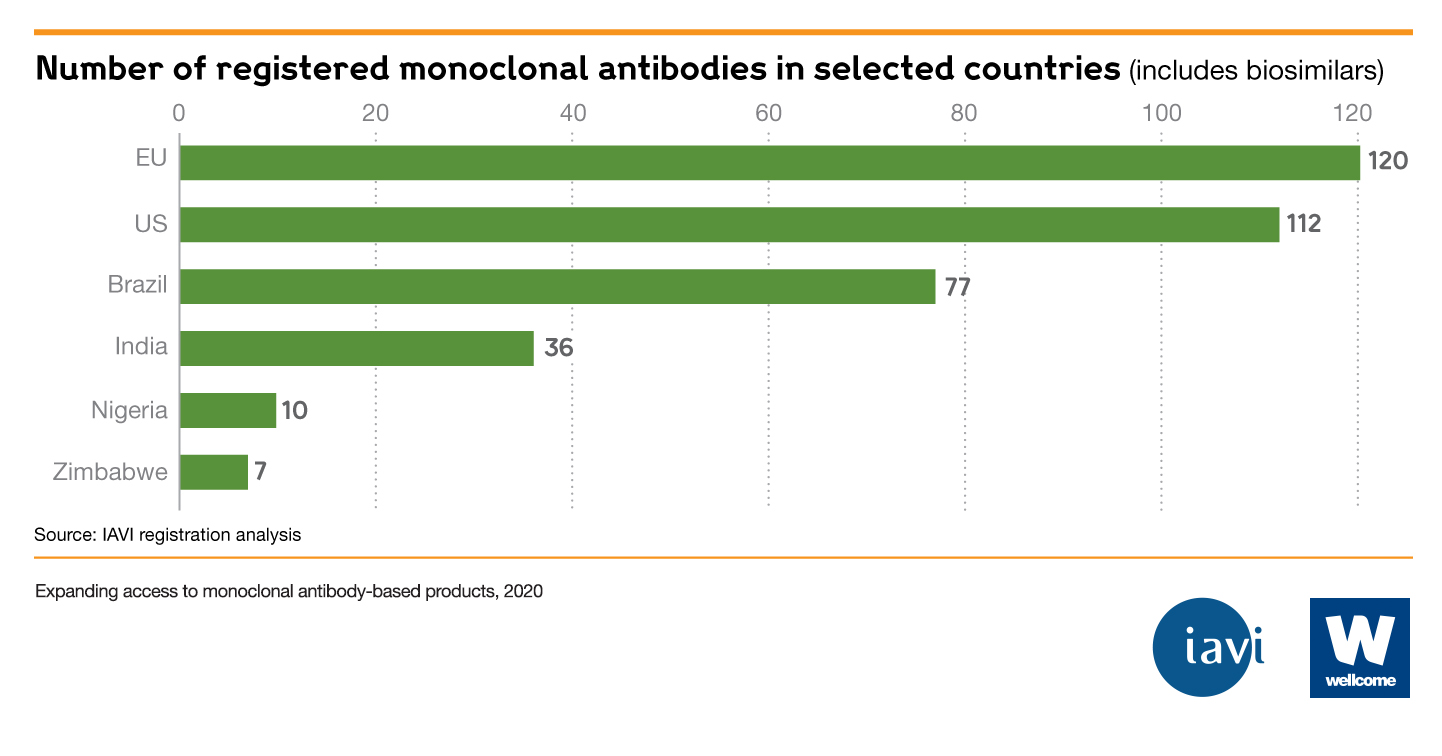
- Few, if any, mAbs are available in low- and middle-income countries (LMICs). India – which has the most registered mAbs for a LMIC – has 36, compared to the US (112) and Europe (120).
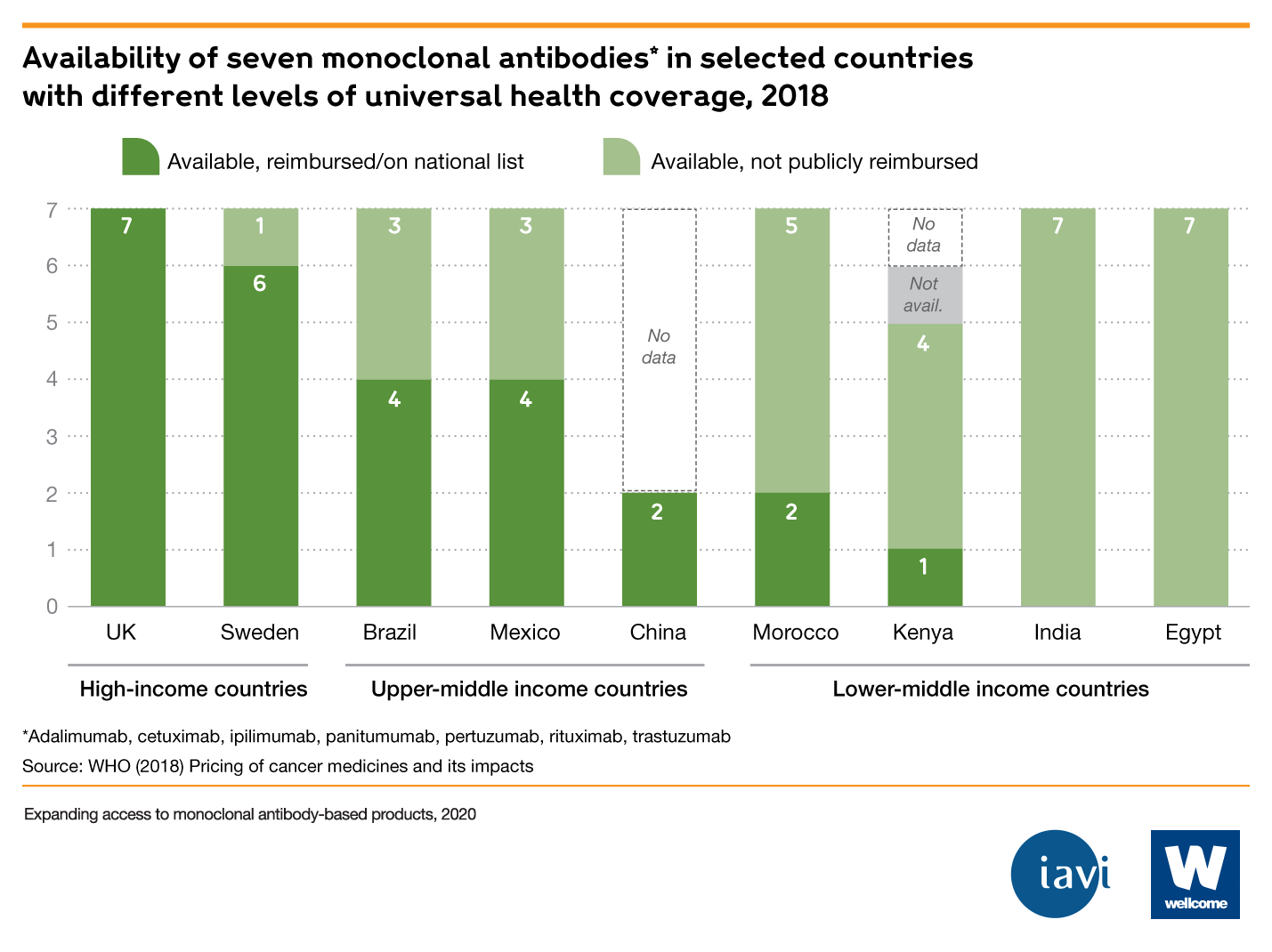
- Monoclonal antibodies are among the world's most expensive treatments. For example, the median price for mAb treatment in the United States is $15k-200k per year. Where mAbs are available in LMICs, they often aren’t reimbursed by public health systems, which adds to their unaffordability.
We need to take action to reshape the monoclonal antibody world
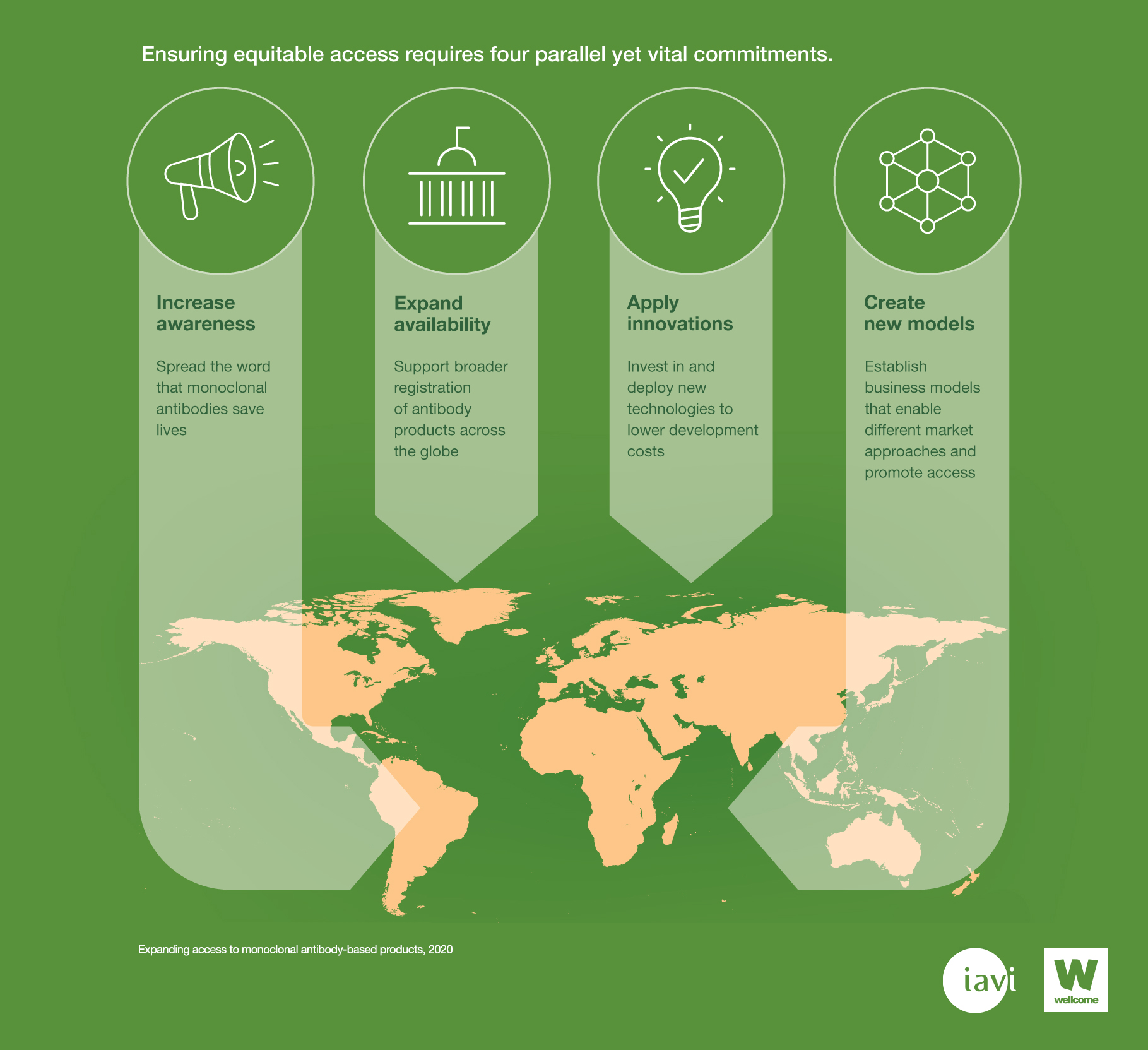
Ensuring equitable access to monoclonal antibodies requires four parallel commitments:
- Increase awareness: spread the word that monoclonal antibodies save lives
- Expand availability: support broader registration of monoclonal antibodies across the globe
- Apply innovations: invest in and deploy new technologies to lower development costs
- Create new models: establish business models that enable different market approaches and promote access.
Group title
Downloads
Group
Listing layout
Link List
Content
Linked file
sarahtucker

File
File size
3686334 bytes
Mime type
application/pdf
Title
Expanding access to monoclonal antibody-based products
Date
Listing text
A roadmap to making lifesaving monoclonal antibody treatments affordable and available, particularly to those living in low- and middle-income countries.
Priority areas
Topics
Teams
Type
Linked file
sarahtucker

File size
1701444 bytes
Mime type
application/pdf
Title
Expanding access to monoclonal antibody-based products: executive summary
Listing text
The key findings from our report which sets out a roadmap to making monoclonal antibodies affordable and available, particularly to those living in low- and middle-income countries.
Type
Linked file
sarahtucker

File
File size
393196 bytes
Mime type
application/pdf
Title
Expanding access to monoclonal antibody-based products: appendix
Listing text
The appendix to our report which sets out a roadmap to making monoclonal antibodies affordable and available, particularly to those living in low- and middle-income countries.
Type
Linked file
sarahtucker

File size
415911 bytes
Mime type
application/pdf
Title
Monoclonal antibodies: a new era in the treatment and prevention of disease
Listing text
What monoclonal antibodies are and how they are used to treat and prevent disease.
Type
Linked file
sarahtucker

File size
203700 bytes
Mime type
application/pdf
Title
The emerging role of monoclonal antibodies in epidemic/pandemic preparedness and response
Listing text
The role that monoclonal antibodies could play in tackling epidemics and pandemics, including COVID-19, Ebola and Nipah.
Type
Linked file
sarahtucker

File
monoclonal-antibodies-supplement-hiv.pdf (54.59 KB)
File size
55903 bytes
Mime type
application/pdf
Title
The development of HIV-specific broadly neutralising antibodies
Listing text
How HIV-specific monoclonal antibodies are being developed, with the aim of reducing infection rates.
Type
Linked file
sarahtucker

File size
212789 bytes
Mime type
application/pdf
Title
Combination monoclonal antibodies and alternate formats
Listing text
How researchers are testing combinations of monoclonal antibodies, and exploring different formats to develop and manufacture them.
Type
Linked file
sarahtucker

File size
44608 bytes
Mime type
application/pdf
Title
India’s biopharmaceutical business: an evolving success story
Listing text
How India has emerged as a global leader in the production of biosimilars, which are lower-priced versions of monoclonal antibodies.
Type
Group
Group title
Infographics
Group
Gallery images
File size
235278 bytes
Image width
1431px
Image height
1056px
Filename
top-10-drugs-by-global-sales-2019.png
Image
Download allowed
On

File size
267975 bytes
Image width
1808px
Image height
1106px
Filename
monoclonal-antibody-market-is-unbalanced_0.png
Image

Download allowed
On
File size
125590 bytes
Image width
1442px
Image height
756px
Filename
number-of-registered-monoclonal-antibodies-in-selected-countries.png
Image
Download allowed
On

File size
180168 bytes
Image width
1251px
Image height
900px
Filename
regulatory-approval-dates-for-monoclonal-antibodies.png
Image
Download allowed
On
File size
514873 bytes
Image width
1512px
Image height
1058px
Filename
concentration-of-global-pharmaceutical-manufacturing.png
Image
Download allowed
On
File size
98443 bytes
Image width
1429px
Image height
1070px
Filename
availability-of-monoclonal-antibodies-in-selected-countries.png
Image
Download allowed
On

File size
874882 bytes
Image width
1819px
Image height
1663px
Filename
four-commitments-to-monoclonal-antibody-equitable-access.png
Image
Download allowed
On
Create hero image
Off
Infographics gallery
Off
Group title
More information
Group
Body
- For more information, contact Lindsay Keir at l.keir@wellcome.org.
Listing layout
Image card with CTA link (Horizontal card)
Content
Body
Innovations Flagships support the development of exciting new products, technologies and other interventions to prevent or treat disease.
Title
Innovations Flagships
Link
/what-we-do/our-work/innovations-flagships
Published date
Modified date
Image

File size
2044543 bytes
Image width
1666px
Image height
1080px
Filename
innovations-flagships-listing-586.jpg
Image

Download allowed
On
Body
Director of Innovation Stephen Caddick explains what flagships are, how they’ll work and what we want to achieve.
Title
Wellcome Flagships will focus on five health innovation challenges
Link
/news/wellcome-flagships-will-focus-five-health-innovation-challenges
Content type
Explainer
Published date
Modified date
Image

Credit
Dieter Telemans/Panos
Caption
Children in the Sankuru River, Democratic Republic of the Congo. One Flagship will focus on drug discovery for neglected tropical diseases, the most common infections for people living in poverty.
File size
1100244 bytes
Image width
1200px
Image height
1200px
Filename
wellcome-flagships-focus-five-health-innovation-challenges-20180905-1200x1200.jpg
Image

Download allowed
On
Body
We want to help the world overcome Covid-19. Research is the best way to save lives – but more investment in accessible treatments, tests and vaccines is urgently needed to change the course of the pandemic.
Title
Coronavirus (Covid-19): supporting global research and development
Link
/what-we-do/our-work/coronavirus-covid-19
Published date
Modified date
Image

File size
866480 bytes
Image width
3333px
Image height
1204px
Filename
coronavirus-covid-19-supporting-global-research-and-development-1600px_0.jpg
Image

Download allowed
On
Published on
Display in page navigation
On
Exclude from listings
Off